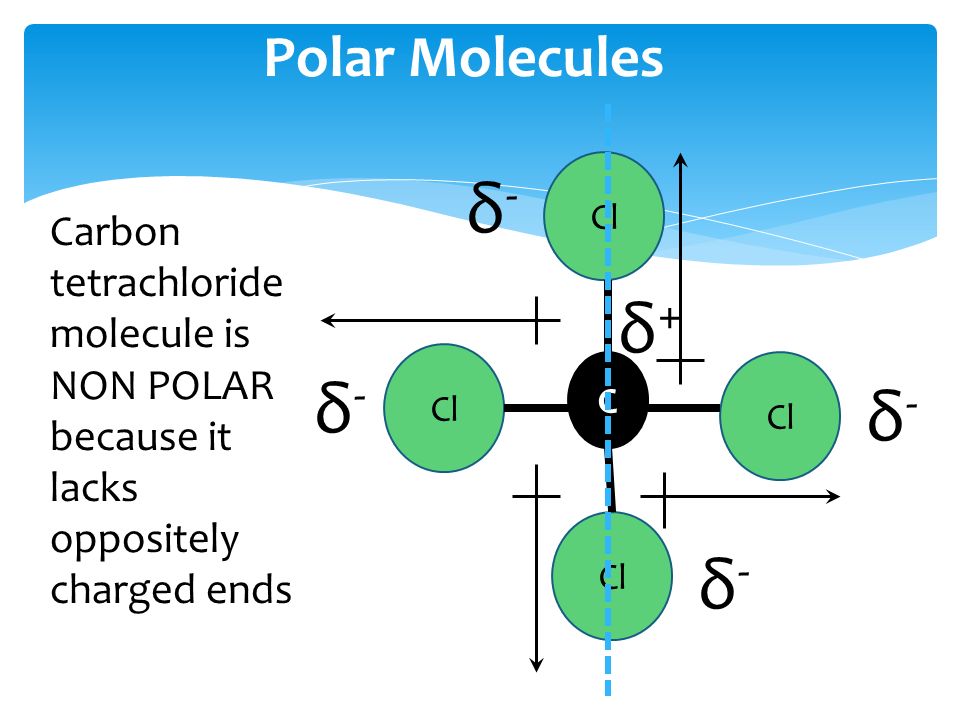
Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar

Explain why carbon tetrachloride molecules are not polar while carbon trichloride is polar - Digital Teachers Uganda

OneClass: The four bonds of carbon tetrachloride (CCI_4) are polar, but the molecule is nonpolar beca...

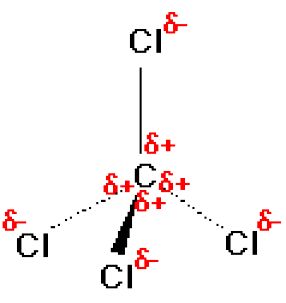
Why is carbon tetrachloride non-polar, while the other tetrachlorides of group four elements are polar? - Quora

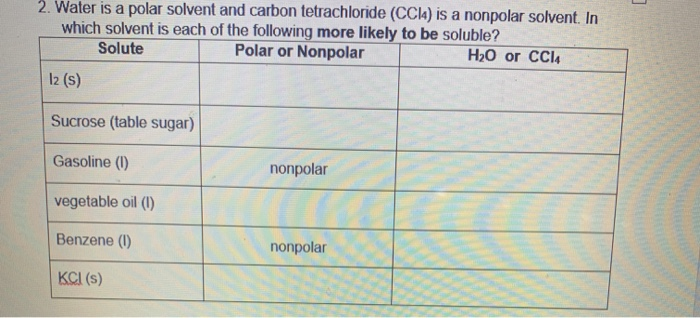
SOLVED: Consider two solvents that are commonly used for organic chemistry reactions: CH2Cl2 and CCl4. Interestingly, studies have shown that one of these solvents is polar and one is non polar. Draw

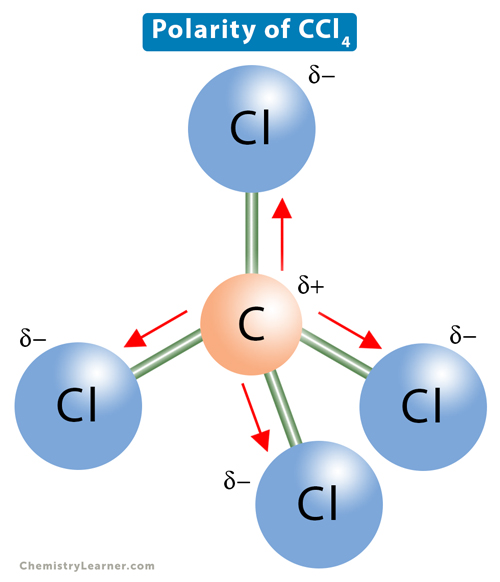
Carbon tetrachloride CCl4 lewis dot structure, molecular geometry, polar or nonpolar, Bond angle | Molecular geometry, Molecular shapes, Molecular

Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar
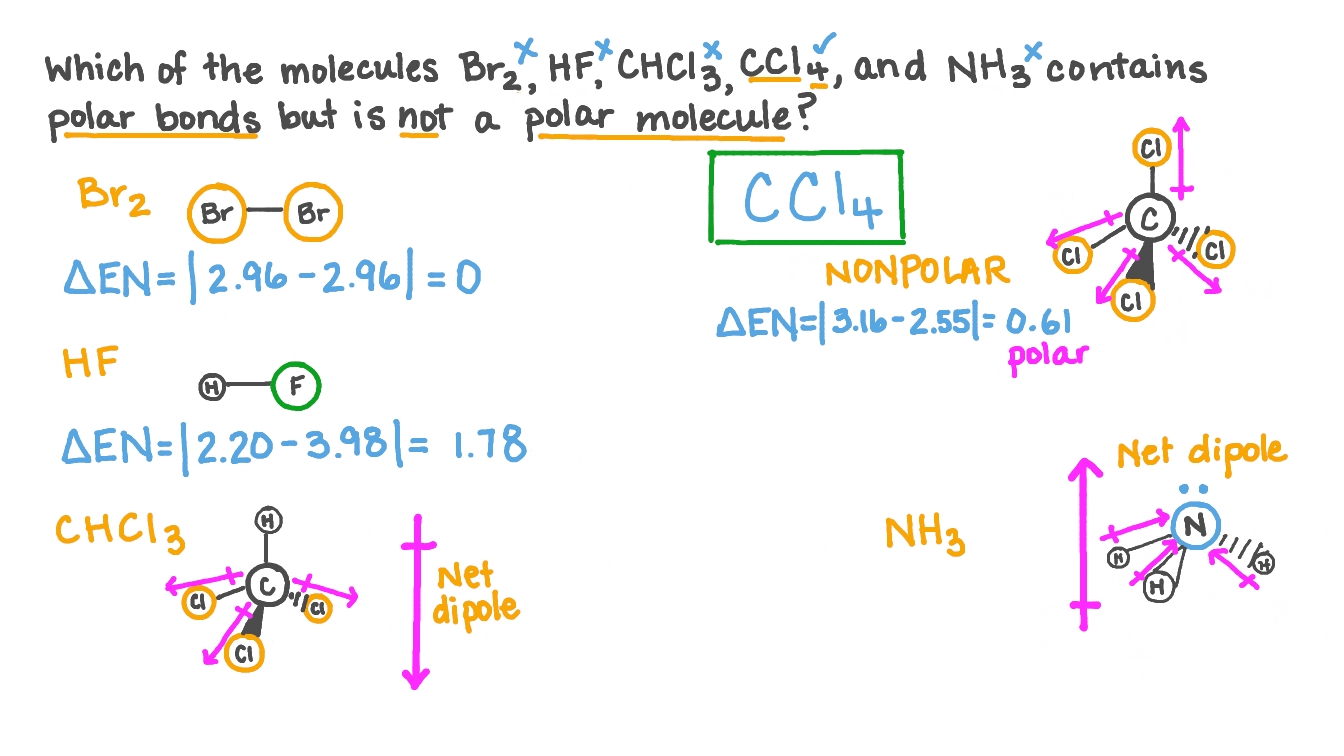
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa